

Levi Olmstead


Pharmaceutical companies are always looking for new opportunities to improve offerings and deliver better products, but traditional ways of R&D no longer work and the pharma industry faces significant digital transformation initiatives.
To remain competitive and keep relevancy in the industry, enterprise pharma companies must modernize legacy applications by migrating to new cloud technologies to have a competitive advantage, provide better quality assurance, create better products, maintain compliance, and continue to grow.
For most pharma companies, cloud transformation is powered by advances in AI, process automation, and big data to improve decision-making and efficiency. But that starts with undergoing a digital transformation.
Digital transformation in pharma is a significant investment — but all companies, regardless of industry, face digitalization challenges.
In this article, we’ll explore the impact of digital transformation on the pharmaceutical and life science industry, examples of digitalization in the industry, and key digital transformation challenges unique to pharma.
Digital transformation in the pharma sector refers to companies updating legacy applications, systems, and processes with cloud applications and emerging technologies to optimize, improve, and revolutionize drug development, manufacturing, distribution, and patient care – as well as enterprise operational functions like sales, procurement, and people management.
Digital transformation initiatives in pharma focus on enhancing operational efficiency, driving innovation, providing better customer experiences, and improving the quality of products and services.
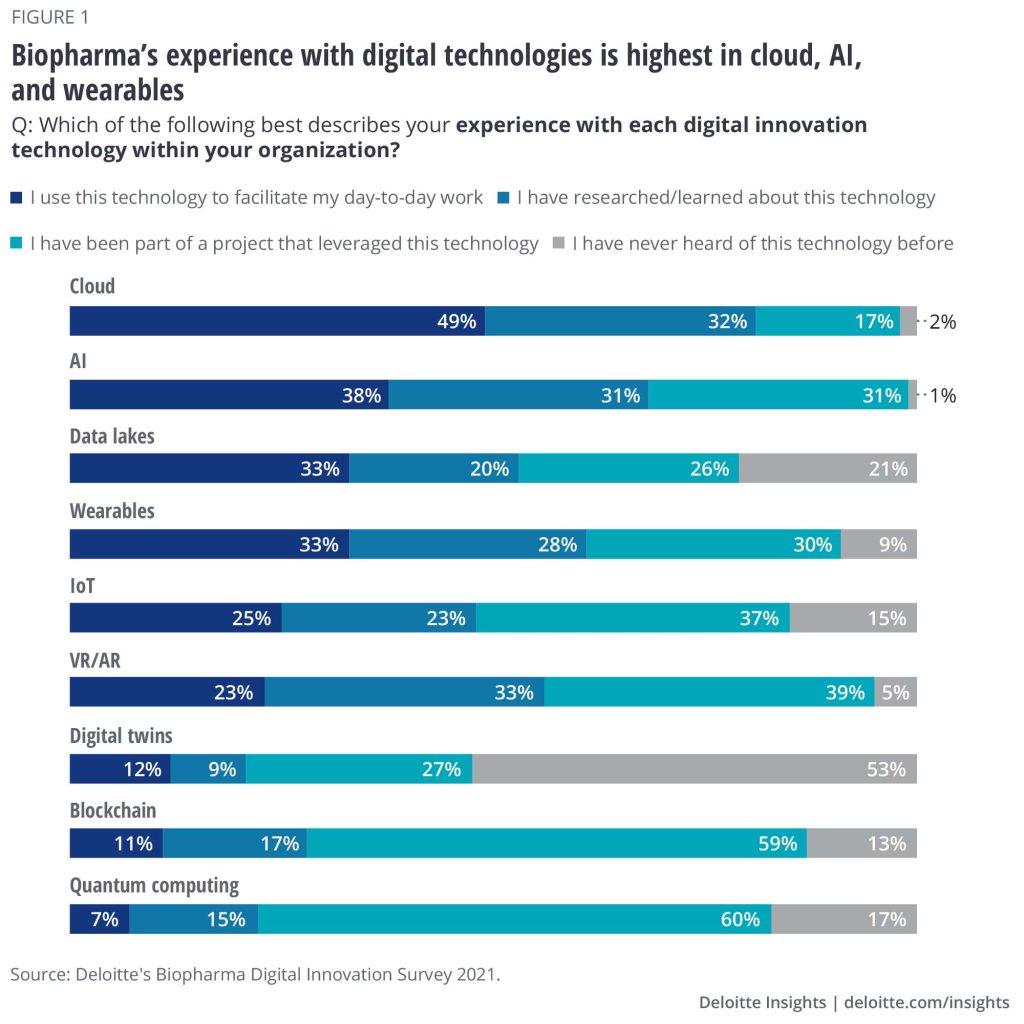
To understand the digital adoption and maturity level of new technologies in pharma, Deloitte surveyed 150 leaders from large biopharma companies of $1B+ revenue. Its results found that certain technologies are widely adopted and integrated into the operations of pharma companies, with cloud (49%), AI (38%), data lakes (33%), and wearables (33%) leading adoption in day-to-day pharma operations.
Here are a few examples of digital transformation initiatives contextual to the pharma industry and life science companies.
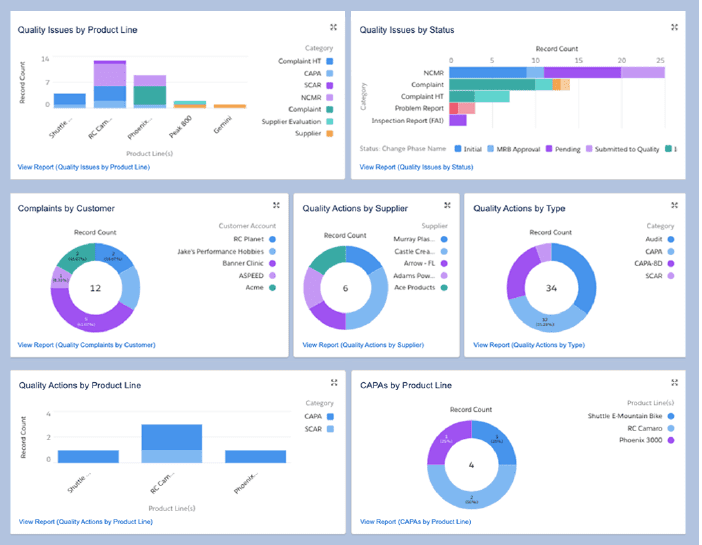
Quality management systems (QMS) use data analytics and real-time monitoring to identify quality issues before they spread quickly. Quickly detecting faulty products — especially in the case of something as sensitive as pharmaceuticals — not only saves money and maintains a high brand standard, but can also save lives.
A QMS provides oversight into several functions related to maintaining quality standards, including:
A regulatory information management (RIM) system centralizes and manages all regulatory data, making it easier to ensure all processes and systems align with compliance requirements. RIM systems digitize the approvals process, reducing the risk of human error and minimizing complexity.
In pharma, a RIM system supports the following processes:
E-clinical platforms can improve the speed and quality of trials while managing costs and resources. It streamlines the collection, analysis, and reporting of clinical trial data, allowing for faster decision-making with lower costs. E-clinical platforms also provide access to real-time data, allowing for in-the-moment changes or adjustments.
Introducing e-clinical platforms to trials can also support pharma companies with:
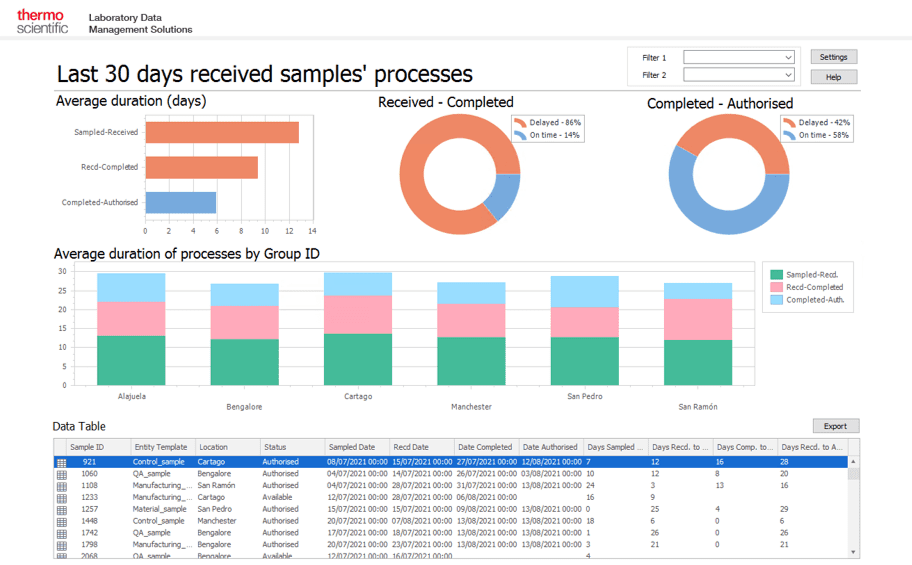
DataOps can improve the integration and management of data, creating a more consistent data environment. In the pharma industry, this means faster insights and improved decision-making that lead to safer and more effective care.
DataOps can support pharma digital transformation by:
Artificial intelligence (AI) and machine learning (ML) can analyze massive datasets in minutes, making it easier to make critical decisions like identifying drug candidates, predicting and forecasting outcomes, and optimizing logistics. Using AI and ML to improve pharma decision-making means better patient care and more positive care outcomes.
AI and ML in pharma can make the following processes more efficient:
Between running trials and providing care via pharmaceuticals, companies must provide those under their care with access to reliable support. Digital tools can give 24/7 access to healthcare info and telemedicine support to ensure their safety and provide the best possible care.
Providing on-demand patient support can:
Digital pills contain an ingestible sensor that allows healthcare providers to monitor patient care and medication efficiency. The pill sensor collects and transmits data that can be used to adjust care plans more effectively.
The use of digital pills can also:
While digital transformation initiatives can dramatically improve the pharma industry, digitalization is a major project that requires dedicated project scoping, budget, resources, migration, implementation, deployment, and ongoing support.
There are also digital transformation challenges that are contextual the the pharma industry. Here are some of the biggest hurdles pharma companies face when undergoing digitization.
Digitization often involves the use of mass amounts of data — and in pharma’s case, that data is linked to patients and their sensitive care information. When dealing with sensitive patient information, it can raise concerns about privacy and data security.
Companies need to ensure they’re complying with regulations like GDPR and HIPAA. Not only can failure to comply lead to steep fines, but a breach of sensitive patient data can lead to a lack of trust and a negative perception of the company.
New digital operations and processes mean employees must know how to work with next-generation tools and technologies, like QMS and RIM systems, among many other new application types.
Pharma employees must be provided proper onboarding and upskill training to do their jobs efficiently with these new digital applications and processes. Without proper training, pharma companies will have employees who can’t use these new technologies – leading to poor end-user performance and productivity, failure to achieve digital transformation ROI, and much more that impacts pharma companies’ bottom line, output, customer experience, and revenue,
Introducing employees to new digital skills may lead to resistance and pushback. Companies must implement effective change management initiatives to ensure they get the most out of their digital transformation.
Introducing new technology requires properly integrating tools across the entire organization, as well as building a compatible IT infrastructure to match the way your departments and teams work. This means integrating and migrating all critical data to the cloud or building a hybrid storage environment, as well as breaking down data silos and enabling your pharma company’s various departments with data warehouses that bring together all this data.
Technology changes can be complex, so you’ll need to build a team capable of handling these problems or hire an outside digital transformation consulting company. Your IT team should be proactive in preventing significant issues like application downtime or lost data – and should be able to provide help desk and service support to team members who need assistance using these new pharma-specific applications.
Pharma IT teams can provide contextual, real-time support to employees through:
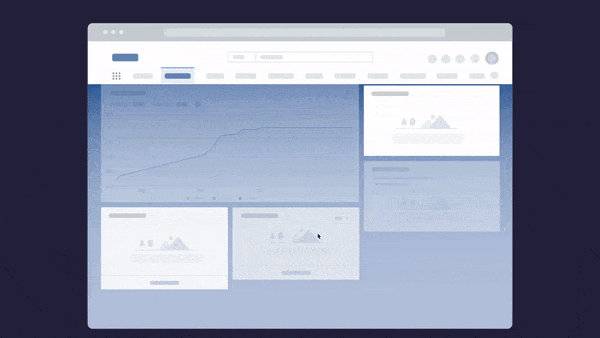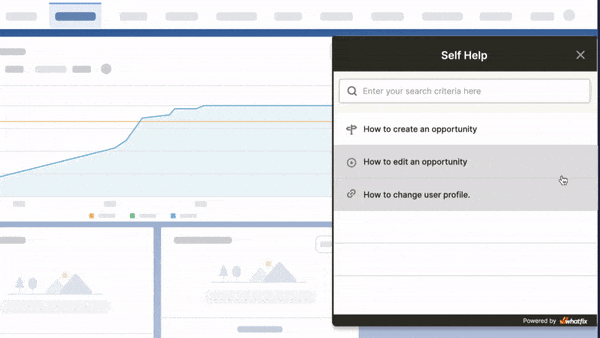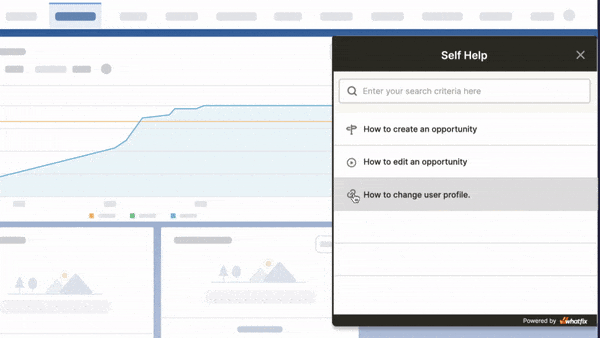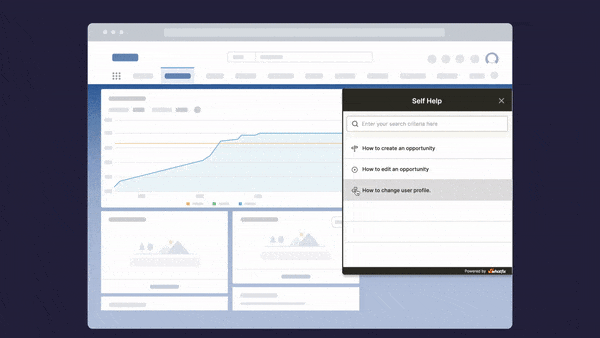
Above: Example of contextual help and user support built with a Digital Adoption Platform.
Whatfix’s Self-Help overlays on to any web application, desktop application, mobile app, or website. It provides contextual help to users and integrates with your FAQs, support center, LMS, user documentation, and more. Users are presented with common issues and help content for their contextual area in the application, or they can use an open-ended search to find the specific help content they’re looking for. These help support cards often prompt in-app guidance, walking users through the specific workflow they need help on.
Even while undergoing a digital transformation, pharma companies must prioritize meeting FDA requirements and other drug regulations. These rules are constantly evolving, but they’re there to maintain patient safety and ensure legal compliance.
While determining what technological developments to implement, look for opportunities to make meeting regulations easier. Put patient care at the center of all the decisions your team makes.
It’s exciting to advance pharma technology as much as possible, but ethics play a crucial role in how technology is used. This is especially true when working with patient data. Regarding research and development, patients’ and trial participants’ safety always needs to come first.
Digital transformation is challenging, costly, resource-intensive, and have lengthy implementation and migration cycles. Accelerate your pharma digital transformation by enabling your employees with contextual in-app guidance and self-help support with Whatfix’s digital adoption platform (DAP)
Whatfix provides moment-of-need, in-the-flow-of-work support and guidance to pharma employees on all new technologies and process changes – helping them to reduce time to proficiency and achieve new levels of productivity and proficiency through better software adoption.
Whatfix also provides product analytics and user behavior tracking to understand how employees are engaging with technologies and workflows, identify areas of user friction or process inefficiencies, map optimal end-user flows for different departments and roles, and drive overall adoption of pharma enterprise systems and technologies like QMS, RIM, Procurement, Contract Management, and more.
Software clicks better with Whatfix's digital adoption platform
Enable your employees with in-app guidance, self-help support, process changes alerts, pop-ups for department announcements, and field validations to improve data accuracy.
Thank you for subscribing!